Click Chemistry Assay for Peptide Coating Evaluation
December 18, 2025
In a pioneering 2025 study published in Advanced Materials Interfaces, Julia Felsch and colleagues introduced an innovative assay platform that synergistically integrates metabolic glycoengineering with click chemistry to efficiently evaluate complement-regulatory peptide coatings on endothelial cells, demonstrating a novel strategy for localized immunomodulation in transplantation medicine. This work establishes a robust methodology where azide-modified sugars are metabolically incorporated into cell-surface glycans, enabling precise, covalent conjugation of factor H-binding peptides via copper-free strain-promoted azide-alkyne cycloaddition (SPAAC), thereby creating a protective coating that effectively impairs alternative pathway complement activation and reduces thromboinflammatory damage. By validating the functional efficacy of these peptide coatings under physiologically relevant conditions—showing significant reduction in C3b opsonization and terminal complement complex deposition on both human and porcine endothelial cells—the research provides a versatile, biocompatible tool for targeted therapeutic screening and biomedical surface engineering.
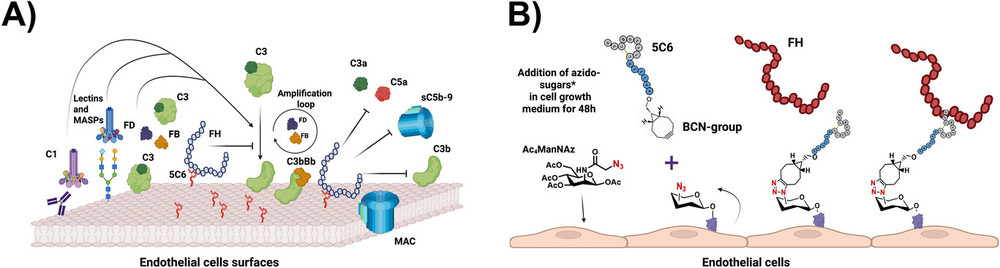 Fig.1 A surface engineering strategy for complement control on endothelial cells via in situ regulator recruitment. (Felsch, et al., 2025)
Fig.1 A surface engineering strategy for complement control on endothelial cells via in situ regulator recruitment. (Felsch, et al., 2025)
Background and Research Objectives
The complement system plays a dual role in immunity: while it defends against pathogens, its dysregulation can lead to tissue damage in conditions like transplantation and ischemia-reperfusion injury (IRI). Endothelial cells are particularly vulnerable, as antibody binding or hypoxia triggers complement activation, resulting in opsonization, inflammation, and cell death. Current therapeutic strategies often involve systemic complement inhibition, which carries risks of immunosuppression. As an alternative, localized surface coatings that recruit natural regulators like factor H (FH) have emerged. FH is a key regulator of the alternative pathway (AP), and peptides such as 5C6 can mimic pathogen strategies to bind FH and suppress complement amplification. However, developing and testing such coatings on living cells requires precise, biocompatible methods. This study addresses this gap by creating a reproducible assay system using metabolic glycoengineering to introduce clickable handles on endothelial glycans, followed by peptide conjugation via click chemistry. The platform enables real-time evaluation of FH recruitment and complement inhibition, with implications for designing protective coatings in biomedical research.
Methodology: Integrating Metabolic Glycoengineering and Click Chemistry
The assay development began with optimizing metabolic glycoengineering for human (HMEC-1 and HUVEC) and porcine (PIEC) endothelial cells. Azido-sugars, including acetylated mannose (Ac4ManNAz) and galactose (Ac4GalNAz) derivatives, were administered to cells at concentrations up to 50 μM. Cell viability assays confirmed minimal toxicity, allowing effective incorporation of azide groups into surface glycans. The accessibility of these groups was verified using a DBCO-modified fluorescent dye (DBCO-AF647), which binds to azides via copper-free click chemistry. Flow cytometry and fluorescence microscopy revealed uniform distribution of azide handles across cell surfaces, with higher incorporation efficiency for Ac4GalNAz in porcine cells.
 Fig.2 Cytotoxicity assessment of azido-sugars on endothelial cells via metabolic activity (XTT assay). (Felsch, et al., 2025)
Fig.2 Cytotoxicity assessment of azido-sugars on endothelial cells via metabolic activity (XTT assay). (Felsch, et al., 2025)
Next, a cyclic FH-binding peptide (5C6) and a scrambled control (scr5C6) were synthesized with N-terminal bicyclononyne (BCN) groups for click conjugation. Coating was performed by incubating azide-modified cells with BCN-5C6, resulting in covalent triazole linkages. A fluorescent derivative (CF-BCN-5C6) was used to visualize peptide distribution, showing even coating across the glycocalyx. Functional assays involved incubating coated cells with purified FH or normal human serum (NHS), with FH binding detected via fluorescent antibodies. The system was tested under physiological conditions, including Mg2+-EGTA serum to restrict complement activation to the AP, mimicking IRI-like environments.
Key Findings and Results
The study demonstrated successful peptide coating and FH recruitment on both human and porcine endothelial cells. Azide incorporation was concentration-dependent, with saturation observed at ~25 μM for Ac4ManNAz in human cells, while porcine cells showed linear responses. Peptide coating via click chemistry achieved uniform surfaces, as confirmed by microscopy, with minimal non-specific binding. FH recruitment assays revealed that 5C6-coated cells bound FH efficiently from purified sources and NHS, in a dose-dependent manner, whereas scr5C6 controls showed negligible binding. Fluorescence microscopy illustrated widespread FH distribution on coated cells, underscoring the coating's effectiveness.
Functional stability tests indicated that azide handles remained accessible for up to 6 hours, with ~50% retention after 24 hours. Peptide coatings maintained FH-recruiting activity for several hours, though a gradual decline was observed, potentially due to proteolysis or internalization. Complement activation assays on PIEC cells exposed to Mg2+-EGTA serum showed that 5C6 coatings significantly reduced C3b/iC3b opsonization and terminal complement complex (TCC) deposition, confirming AP inhibition. Interestingly, even low peptide densities sufficed for robust complement control, suggesting high efficacy of FH recruitment.
Discussion and Implications
This assay platform offers a robust method for screening complement-regulatory coatings, with several advantages: (1) It uses biocompatible, click chemistry-based conjugation that minimizes cell damage; (2) It allows real-time monitoring of peptide performance under physiologically relevant conditions; and (3) It is adaptable to various cell types, enabling broader applications in xenotransplantation and autoimmune disease research. The findings highlight the potential of peptide-based strategies to locally modulate complement activity, reducing reliance on systemic drugs. However, challenges remain, such as coating stability in vivo and translation to whole-organ settings. Future work could explore optimized peptide derivatives with enhanced proteolytic resistance or in combination with genetic engineering for transgenic expression.
Connection to GlycoCLICK™ Technology at CD BioGlyco
The methodologies employed in this study resonate deeply with the core services of CD BioGlyco's GlycoCLICK™ Platform. GlycoCLICK™ specializes in click chemistry-based solutions for glycobiology, including custom synthesis of glycoconjugates, surface modifications, and biomaterial preparation. For instance, the GlycoCLICK™-based Modification Service and GlycoCLICK™-based Labeling Service offer tailored approaches for introducing functional groups like azides and alkynes into biological systems, similar to the metabolic glycoengineering used here. Additionally, the GlycoCLICK™-based Drug Development Service could facilitate the optimization of FH-recruiting peptides for therapeutic use, leveraging high-throughput screening and expert support.
Researchers interested in advancing such assay systems or developing related applications can benefit from CD BioGlyco's comprehensive portfolio. The company provides GlycoCLICK™-based Chemical Synthesis Services for peptide and glycan production, as well as GlycoCLICK™-based Biomaterial Preparation Services for creating functionalized surfaces. By collaborating with CD BioGlyco, scientists gain access to advanced platforms and experienced professionals, ensuring high-quality results in glycobiology projects.
Conclusion
This study establishes a versatile assay system for evaluating complement-regulatory peptide coatings, highlighting the synergy between metabolic glycoengineering and click chemistry. The platform not only validates the efficacy of FH-recruiting strategies but also provides a foundation for future innovations in immune modulation. For researchers seeking to leverage these techniques, CD BioGlyco's GlycoCLICK™ services offer reliable support, from custom synthesis to functional testing.
Reference
- Felsch, J.; et al. Assay system based on metabolic glycoengineering-enabled click chemistry for the efficient evaluation of complement-regulatory peptide coatings on endothelial cells. Advanced Materials Interfaces. 2025, 12(19): e00350.

 Fig.1 A surface engineering strategy for complement control on endothelial cells via in situ regulator recruitment. (Felsch, et al., 2025)
Fig.1 A surface engineering strategy for complement control on endothelial cells via in situ regulator recruitment. (Felsch, et al., 2025) Fig.2 Cytotoxicity assessment of azido-sugars on endothelial cells via metabolic activity (XTT assay). (Felsch, et al., 2025)
Fig.2 Cytotoxicity assessment of azido-sugars on endothelial cells via metabolic activity (XTT assay). (Felsch, et al., 2025)