Lectin Inhibition with Click-Derived Glycocyclopeptides
December 18, 2025
Introduction to Glycocyclopeptide Research
The field of glycobiology has witnessed significant advances in the development of glycoclusters—synthetic molecules that display multiple carbohydrate residues for studying carbohydrate-protein interactions. A groundbreaking study by Bossu et al. (2011) published in the Journal of Carbohydrate Chemistry presents a comparative analysis of two efficient strategies for constructing tetravalent glycocyclopeptides: oxime ligation and copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC). This research not only provides valuable insights into synthetic methodology but also demonstrates the importance of linkage chemistry in biological recognition processes.
At CD BioGlyco, we recognize the critical importance of such methodological advances, which is why we have developed our extensive GlycoCLICK™ technology platform to support researchers in overcoming synthetic challenges in glycoconjugate development.
Research Methodology and Technical Approach
The study employed a systematic comparison between oxime ligation and CuAAC strategies for assembling tetravalent glycocyclopeptides. The researchers synthesized cyclopeptide scaffolds displaying either aldehyde groups (for oxime ligation) or azide functionalities (for CuAAC). These scaffolds were then conjugated with various glycosyl derivatives, including β-galactopyranosyl (βGal), α-mannopyranosyl (αMan), and α-fucopyranosyl (αFuc) residues.
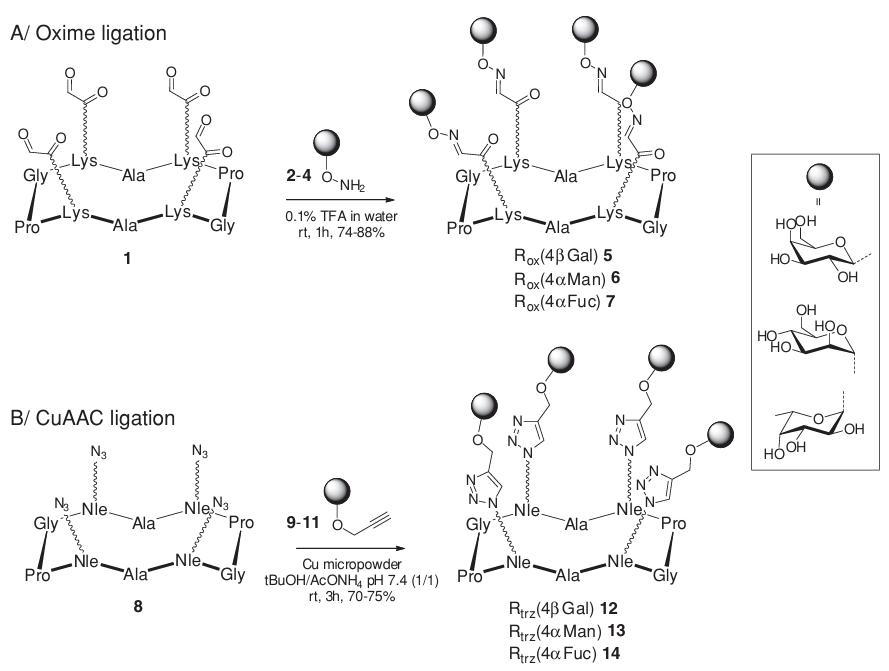 Fig.1 Convergent synthetic strategies for tetravalent glycoclusters. (Bossu, et al., 2011)
Fig.1 Convergent synthetic strategies for tetravalent glycoclusters. (Bossu, et al., 2011)
The oxime ligation procedure was completed within 1 hour in aqueous acidic buffer, yielding glycoclusters 5-7 with 74-88% efficiency after HPLC purification. The CuAAC approach required 3 hours in tert-butanol/sodium acetate buffer with copper micropowder catalysis, producing glycoclusters 12-14 with 70-75% yield. Both methods demonstrated excellent efficiency and reproducibility, with comprehensive characterization confirming the expected structures through NMR spectroscopy and mass spectrometry.
Key Findings and Biological Significance
The most significant finding emerged from the lectin binding studies using enzyme-linked lectin assays (ELLA). While both synthetic approaches produced glycoclusters with similar efficiency, their biological recognition properties differed markedly:
- For Concanavalin A (ConA, specific for αMan), both oxime-linked (6) and triazole-linked (13) glycoclusters showed comparable inhibitory potency (IC50 ≈ 76-79 μM), indicating minimal influence of the linkage chemistry on mannose recognition.
- For Ulex europaeus lectin (UEA-I, specific for αFuc), a dramatic difference was observed. The oxime-linked fucosylated cluster (7) exhibited significantly enhanced inhibition (IC50 = 1 μM) compared to its triazole-linked counterpart (14, IC50 = 16 μM), representing a more than 15-fold improvement in binding affinity.
This finding suggests that the oxime linker may provide favorable spatial orientation or secondary interactions with the UEA-I binding site, highlighting the importance of linkage chemistry in glycocluster design for specific biological applications.
Connection to CD BioGlyco Services
The methodologies explored in this research align perfectly with the comprehensive service portfolio offered by CD BioGlyco through our GlycoCLICK™ platform. Our expertise encompasses:
We specialize in the synthesis of complex glycoconjugates using both CuAAC and other click chemistry approaches, building on the foundational work demonstrated in this study.
Our platform offers sophisticated modification strategies for proteins, surfaces, and biomaterials, enabling precise control over glycosylation patterns and linkage chemistry.
We provide advanced labeling solutions for cells, antibodies, and biomolecules, incorporating the latest developments in glycoconjugate chemistry.
Leveraging insights from lectin binding studies, we support the development of carbohydrate-based therapeutics with optimized binding properties.
Technical Advantages and Innovation
The research by Bossu et al. demonstrates several technical advantages that resonate with CD BioGlyco's approach:
Both oxime and CuAAC strategies allow for efficient assembly of glycoclusters from building blocks, enabling rapid exploration of structure-activity relationships.
These chemoselective ligation methods permit the incorporation of carbohydrates without protecting group manipulations, streamlining synthetic workflows.
The demonstration that linkage chemistry influences lectin binding underscores the importance of methodological choices in glycoconjugate design—a principle we incorporate into our service offerings.
Research Implications and Future Directions
This study opens several avenues for future research that align with CD BioGlyco's expertise:
- Structure-activity relationships: Further investigation into how different linkers influence carbohydrate presentation and recognition.
- Multivalent display optimization: Refinement of glycocluster architectures for enhanced biological activity.
- Therapeutic applications: Development of glycoconjugates for targeted drug delivery and vaccine development.
CD BioGlyco stands ready to support researchers in exploring these directions through our comprehensive technology platform and expert team.
Why Choose CD BioGlyco?
Our company combines the methodological rigor demonstrated in studies like Bossu et al. with practical expertise in glycobiology applications. We offer:
- Advanced technological platform: State-of-the-art facilities for glycoconjugate synthesis and characterization.
- Expert team: Scientists with deep knowledge of carbohydrate chemistry and biological applications.
- Customized solutions: Tailored approaches to address specific research challenges.
- Quality assurance: Rigorous quality control measures to ensure reliable results.
Conclusion
The research by Bossu et al. exemplifies the sophisticated approaches needed to advance glycobiology. By demonstrating the differential effects of linkage chemistry on lectin binding, this study provides valuable insights for designing glycoconjugates with optimized biological properties. CD BioGlyco builds upon such foundational research to offer comprehensive solutions for glycobiology challenges, leveraging our GlycoCLICK™ platform to support innovative research and development.
For researchers interested in exploring glycocyclopeptide synthesis or other glycoconjugate applications, CD BioGlyco offers the expertise and technological resources to accelerate your project. Visit our website to learn more about our services or contact our team to discuss how we can support your research objectives.
Reference
- Bossu, I.; et al. Synthesis of glycocyclopeptides by click chemistry and inhibition assays with lectins. Journal of Carbohydrate Chemistry. 2011, 30(7-9): 458-468.

 Fig.1 Convergent synthetic strategies for tetravalent glycoclusters. (Bossu, et al., 2011)
Fig.1 Convergent synthetic strategies for tetravalent glycoclusters. (Bossu, et al., 2011)