Click-Based Chiral Selector for HPLC
December 18, 2025
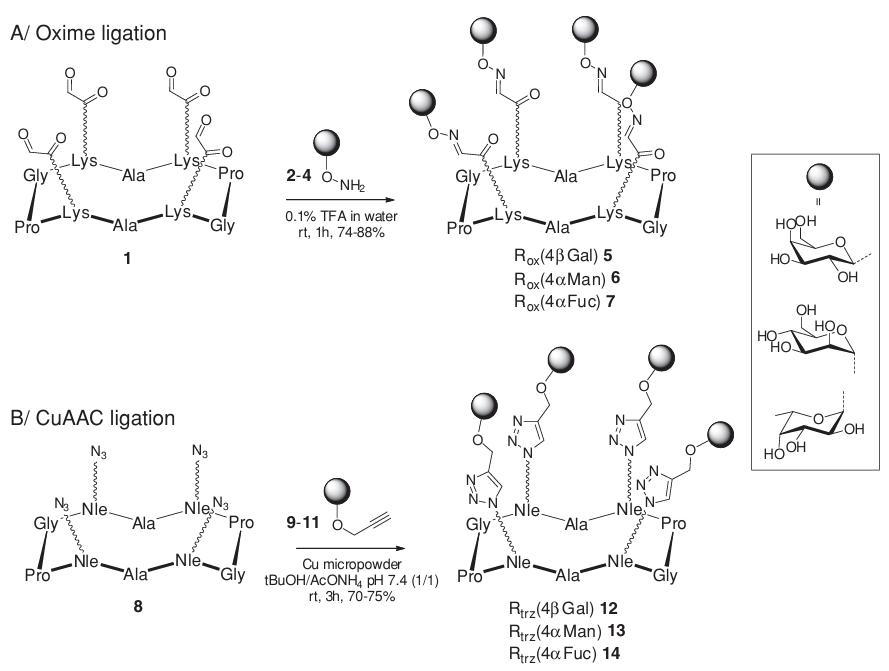
In a groundbreaking study published in the Journal of Carbohydrate Chemistry in 2011, Ivan Bossu and colleagues systematically compared two powerful click chemistry strategies—oxime linkage and copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC)—for the efficient synthesis of tetravalent glycocyclic peptides, revealing the profound impact of linkage chemistry on lectin binding specificity. While both methods demonstrated high synthetic efficiency (70-88% yield) in constructing multivalent glycoclusters containing galactose, mannose, and fucose residues, their biological evaluation revealed a significant difference: the oxime-linked fucoidylated glycoclusters exhibited more than 15-fold higher inhibitory activity (IC50 = 1 μM) against European broom lectin I compared to their triazole-linked counterparts. This demonstrates that the choice of linkage chemistry is not merely a synthetic issue but an important factor determining biological activity. This fundamental work underscores the importance of strategic approach selection in glycoconjugate design, a principle central to the professional services offered by CD BioGlyco through the GlycoCLICK™ platform. The platform utilizes precise chemical methods to develop highly specific lectin inhibitors and therapeutic glycoconjugates.
Background: The Importance of Chiral Separation and Click Chemistry
Chiral molecules, existing as enantiomers, often exhibit distinct biological activities, making their separation imperative for safety and efficacy in drugs and chemicals. HPLC with CSPs is a preferred method, relying on chiral selectors that interact differentially with enantiomers. Cellulose, a natural homochiral polymer, has been derivatized into phenyl carbamates (e.g., 3,5-dichlorophenyl carbamate) to enhance interactions, but early CSPs used physical coating onto silica supports. While effective, these coated phases are prone to leaching in polar solvents like tetrahydrofuran (THF) or chloroform, limiting mobile phase choices and causing column degradation. Covalent immobilization addresses this by chemically bonding the selector to the support, ensuring solvent resistance and longevity. Click chemistry, particularly the Cu(I)-catalyzed azide-alkyne cycloaddition (CuAAC), offers a mild, efficient route for such immobilization, with high specificity and yield. This method has gained traction in glycobiology for constructing complex molecules, as highlighted by CD BioGlyco's GlycoCLICK™ services, which specialize in applications ranging from glycopolymer synthesis to biomaterial preparation.
Summary of the Key Literature Findings
- Introduction and Objectives
The study by Lehrhofer et al. aims to overcome the drawbacks of coated CSPs by developing a covalently immobilized cellulose-based selector using alkyne-azide click chemistry. Focusing on cellulose 3,5-dichlorophenyl carbamate, a proven chiral selector, the team incorporated propargyl anchor groups for covalent attachment to azide-functionalized silica. The objectives included characterization of the selector, evaluation of separation performance under normal-phase conditions, and assessment of solvent compatibility. This approach mirrors the precision and innovation central to CD BioGlyco's ethos, where we support similar advancements through our GlycoCLICK™-based Chemical Synthesis Service.
The synthesis began with microcrystalline cellulose, which was functionalized in a homogeneous DMAc/LiCl/pyridine system. A stepwise carbamoylation strategy was employed: first, 3,5-dichlorophenyl isocyanate was added to quench residual water, followed by 4-propargyloxy-3,5-dichlorophenyl isocyanate to introduce alkyne groups (degree of substitution, DS = 0.45 for the anchor). Excess 3,5-dichlorophenyl isocyanate then achieved near-complete derivatization (total DS ≈ 2.80). The resulting chiral selector (CS1) was characterized using ATR-FTIR, NMR spectroscopy, and elemental analysis, confirming the successful incorporation of anchor groups.
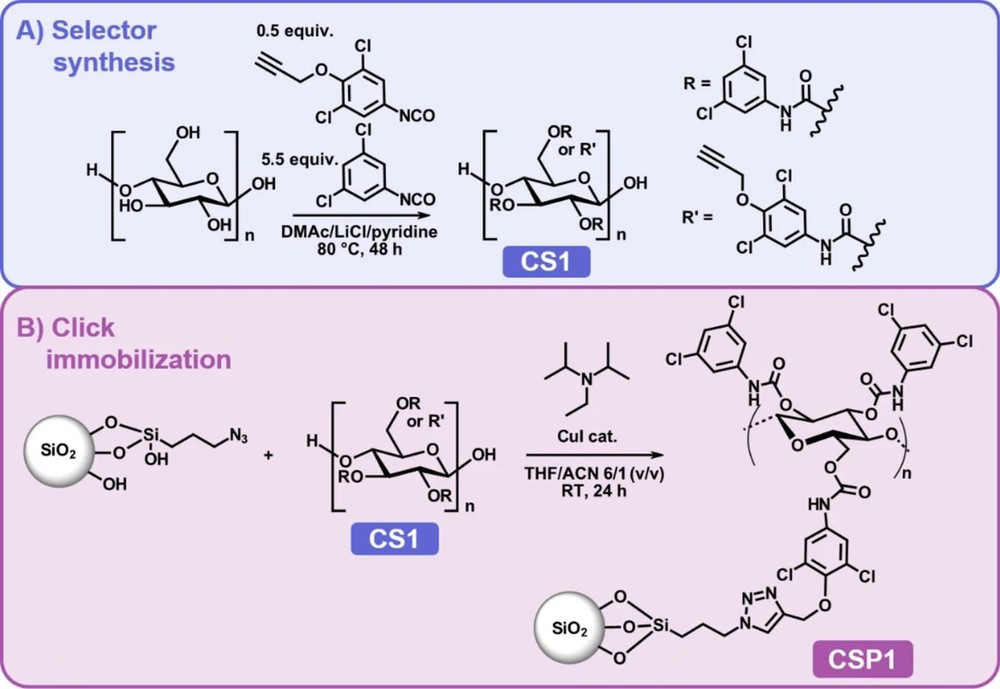 Fig.1 Synthesis of cellulose derivative CS1 via phenyl carbamoylation. (Lehrhofer, et al., 2025)
Fig.1 Synthesis of cellulose derivative CS1 via phenyl carbamoylation. (Lehrhofer, et al., 2025)
For immobilization, silica gel was functionalized with 3-azidopropyl groups (AzPS) via chloropropyl modification and azide substitution. The click reaction between CS1's alkyne groups and AzPS's azide groups, catalyzed by CuI in THF, yielded a covalently bonded CSP (CSP1) with approximately 9 wt% selector loading. Control phases included coated CSPs with 9 wt% (CSP2) and 20 wt% (CSP3) loadings for comparison. All materials were packed into HPLC columns and tested with a set of 16 racemic analytes, including compounds like benzoin and Tröger's base, under normal-phase conditions (n-hexane/i-PrOH 90:10, v/v).
- Results and Performance Evaluation
The immobilized CSP1 exhibited superior enantioseparation compared to coated counterparts. Under standard conditions, CSP1 achieved baseline separation for analytes like 2-phenylcyclohexanone (analyte A) with a resolution (Rs) of 1.50, whereas CSP2 only partially resolved it (Rs = 0.97). Higher loading in CSP3 improved resolution but increased retention times, highlighting a trade-off. CSP1's performance was attributed to the covalent linkage, which preserved the selector's flexibility and accessibility, facilitating better chiral recognition. CSP1 resolved analytes that coated phases struggled with, such as benzoin (B) and Pirkle's alcohol (D).
Solvent compatibility tests demonstrated CSP1's robustness. With mobile phases containing up to 20% strong solvents like ethyl acetate, THF, or chloroform, CSP1 maintained or even enhanced separation for specific analytes. For instance, adding 5% THF boosted the resolution of an ibuprofen derivative (analyte O) from 3.95 to 6.47. Long-term stability assays, involving 33-hour flushing with chloroform-containing eluents, showed minimal performance degradation, underscoring the immobilization's durability.
- Conclusions and Implications
The study concludes that click chemistry provides a reliable method for covalent immobilization, yielding CSPs with broad solvent compatibility and high efficiency. The strategic use of a propargyl anchor with a spacer enhanced flexibility, addressing prior concerns about rigidity in cross-linked systems. This work paves the way for advanced CSP designs and aligns with trends in green chemistry by reducing waste from column bleeding. For researchers, it emphasizes the value of tailored immobilization strategies, a domain where CD BioGlyco's GlycoCLICK™-based Modification Service excels, offering protein modification, surface functionalization, and more.
Connecting to CD BioGlyco's GlycoCLICK™ Services
At CD BioGlyco, we recognize the transformative potential of click chemistry in glycobiology. Our GlycoCLICK™ platform is designed to support innovations like those in Lehrhofer et al.'s study, providing end-to-end solutions for carbohydrate-based research. Here's how our services align with this literature and can benefit your projects:
We specialize in synthesizing complex glycoconjugates using azide-alkyne cycloaddition, similar to the selector development in the study. Our expertise ensures high-purity compounds for CSP applications or drug discovery.
Just as the study immobilized selectors on silica, we offer nanoparticle preparation, hydrogel development, and surface modification to create stable, functional materials for chromatography or drug delivery.
With capabilities in enzyme inhibitor development (e.g., glycosidase inhibitors) and lectin ligand design, we help translate chiral separation insights into therapeutic advances, leveraging click chemistry for precision.
For rapid optimization of CSPs or analytes, our screening services accelerate R&D, reducing time and costs associated with method development.
Our team of glycobiology experts, backed by state-of-the-art platforms, ensures reliable data and customized support. Whether you're developing novel CSPs or exploring glycans, CD BioGlyco is your trusted partner.
Call to Action: Elevate Your Research with CD BioGlyco
The study by Lehrhofer et al. underscores the critical role of click chemistry in advancing chiral separation technologies. At CD BioGlyco, we build on such foundations to offer cutting-edge services that drive progress in glycobiology. Visit our GlycoCLICK™ portal to learn about our full range of services, from glycopolymer synthesis to vaccine development.
This article is based on the publication "Covalent anchoring of a cellulose per(phenyl carbamate) chiral selector onto silica gel through alkyne-azide click chemistry and its utilization" (Cellulose, 2025). For more details, refer to the original paper. CD BioGlyco is not affiliated with the authors but supports such innovative research through its services.
Reference
- Lehrhofer, A.F.; et al. Covalent anchoring of a cellulose per (phenyl carbamate) chiral selector onto silica gel through alkyne-azide click chemistry and its utilization in HPLC. Cellulose. 2025, 1-15.

 Fig.1 Synthesis of cellulose derivative CS1 via phenyl carbamoylation. (Lehrhofer, et al., 2025)
Fig.1 Synthesis of cellulose derivative CS1 via phenyl carbamoylation. (Lehrhofer, et al., 2025)