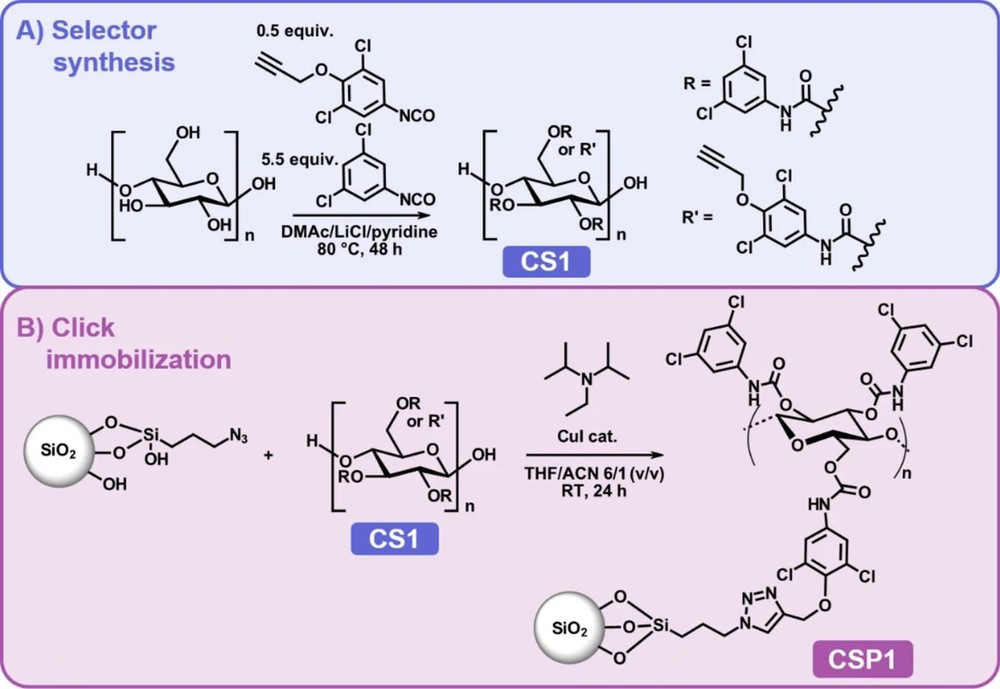
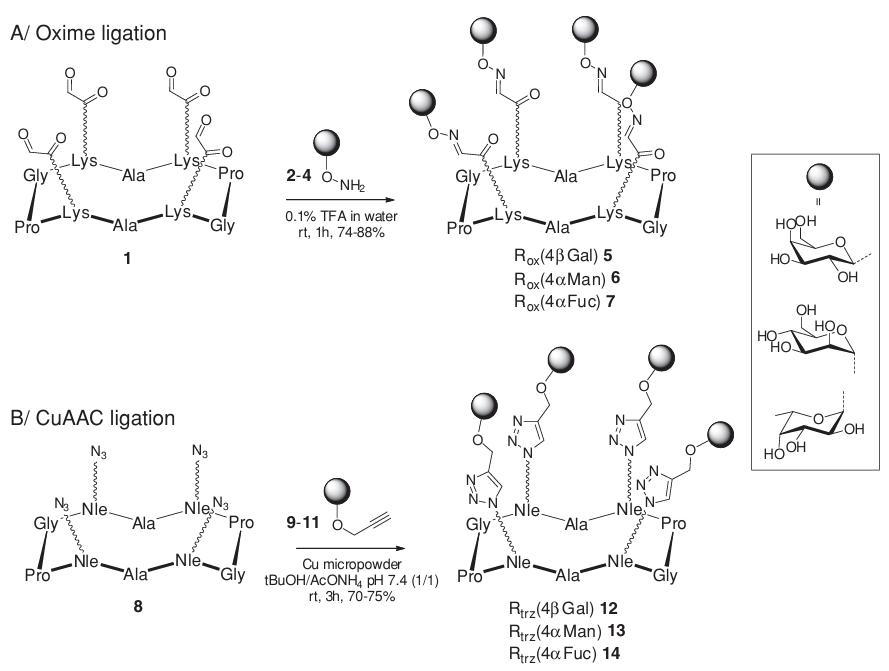
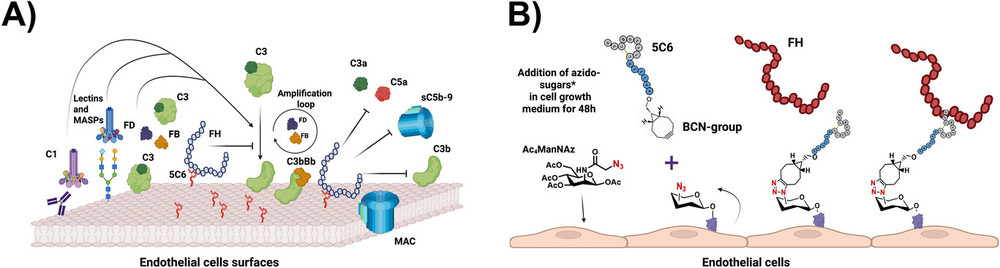
New Frontiers with Metal-Free Click Chemistry
November 18, 2025
A recent review published in Bioconjugate Chemistry titled "Exploring metal-free click reactions: new frontiers in glycochemistry and bioconjugation" delves into the transformative role of metal-free click chemistry in this domain. This comprehensive article, authored by Ramírez-López et al. (2025), highlights four key reactions—thiol–ene coupling (TEC), strain-promoted azide–alkyne cycloaddition (SPAAC), inverse electron-demand Diels–Alder (IEDDA) reaction, and sulfur(VI) fluoride exchange (SuFEx)—that offer selective, efficient, and biocompatible tools for glycoconjugate synthesis under mild conditions. As these methodologies gain traction, CD BioGlyco is leveraging similar principles through our GlycoCLICK™ services to provide cutting-edge solutions for researchers. This article will summarize the critical insights from the review and explore how these innovations align with professional services available for glycobiology applications.
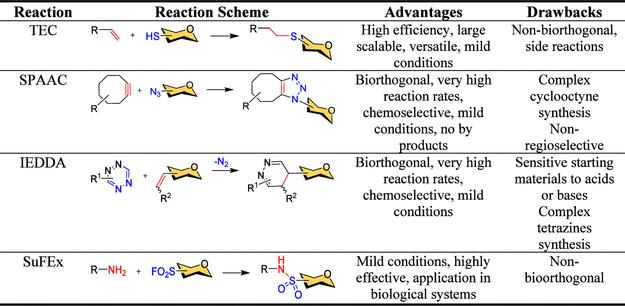 Fig. 1. Comparative analysis of metal-free click reactions. (Ramírez-López, et al., 2025)
Fig. 1. Comparative analysis of metal-free click reactions. (Ramírez-López, et al., 2025)
Introduction to Metal-Free Click Chemistry in Glycoconjugation
Glycoconjugates, including glycoproteins, glycolipids, and proteoglycans, play pivotal roles in cellular recognition, signaling, and immune responses. Abnormal glycosylation is linked to diseases such as cancer, infections, and immune disorders, making glycoconjugates prime targets for therapeutic and diagnostic development. Traditional synthesis methods often involve harsh conditions incompatible with biological systems. The introduction of click chemistry by Sharpless in 2001 revolutionized this field by providing highly selective and efficient reactions. However, metal-catalyzed versions like copper-catalyzed azide–alkyne cycloaddition (CuAAC) face limitations due to copper's cytotoxicity. This has spurred interest in metal-free alternatives, which are not only biocompatible but also bioorthogonal—capable of proceeding in living organisms without disrupting native processes. The review emphasizes how TEC, SPAAC, IEDDA, and SuFEx address these challenges, enabling applications from live-cell imaging to vaccine development.
Thiol–Ene Coupling (TEC): A Versatile Tool for Glycoconjugate Synthesis
TEC, also known as hydrothiolation, involves the radical-mediated addition of thiols to alkenes, forming stable thioethers under mild, often aqueous conditions. Driven by visible light, this reaction is atom-economical and compatible with diverse functional groups. The review outlines its mechanism, where a thiyl radical adds to an alkene in an anti-Markovnikov fashion, followed by hydrogen abstraction to yield the product. A key advantage is its regio- and stereoselectivity, making it ideal for synthesizing S-linked disaccharides and glycopeptides.
For instance, Dondoni and colleagues demonstrated the synthesis of S-linked disaccharides via TEC between thioglycosides and glycals, achieving high yields and diastereoselectivity. This approach has been extended to glycopeptides and glycoproteins; for example, Davis et al. site-specifically ligated glycosyl thiols to olefinic proteins, enabling the preparation of glycoconjugate vaccines. Additionally, TEC facilitates the creation of glycodendrimers and glycoclusters using scaffolds like polyhedral oligosilsesquioxanes (POSS) and calixarenes, enhancing multivalent interactions for biological recognition. Despite its utility, TEC is not bioorthogonal due to potential interference with cellular thiols, limiting in vivo applications.
Strain-Promoted Azide–Alkyne Cycloaddition (SPAAC): Bioorthogonal Labeling and Beyond
SPAAC emerged as a bioorthogonal alternative to CuAAC, involving cycloaddition between azides and strained cyclooctynes without metal catalysts. This reaction offers fast kinetics, high chemoselectivity, and biocompatibility, though it may produce regioisomers. The review highlights SPAAC's role in metabolic glycoengineering, where azide-functionalized sugars are incorporated into cell-surface glycans and labeled with cyclooctyne-conjugated probes for imaging.
Bertozzi's group pioneered this for visualizing sialic acids in live cells and zebrafish embryos, enabling dynamic monitoring of glycan trafficking. SPAAC also underpins therapeutic advances, such as the synthesis of glycofullerenes for antiviral strategies and lysosome-targeting chimeras (LYTACs) for protein degradation. In one example, glycopolypeptides conjugated to antibodies via SPAAC facilitated targeted degradation of extracellular proteins. Moreover, integration with biocatalysis—termed "bioclick chemistry"—has enabled the development of antibody–drug conjugates (ADCs) with improved homogeneity. These applications demonstrate SPAAC's versatility in chemical biology and drug development.
Inverse Electron-Demand Diels–Alder (IEDDA) Reaction: Rapid and Selective Ligation
The IEDDA reaction is a bioorthogonal cycloaddition between electron-deficient dienes (e.g., tetrazines) and electron-rich dienophiles (e.g., strained alkenes), proceeding rapidly under aqueous conditions. Its high efficiency and selectivity make it suitable for metabolic labeling and hydrogel formation. The review cites its use in metabolic glycoengineering, where cyclopropene-modified sialic acids are incorporated into glycans and detected with tetrazine probes.
Additionally, IEDDA enables the fabrication of injectable polysaccharide-based hydrogels for drug delivery and tissue engineering. For example, functionalization of hyaluronic acid with tetrazines and dienophiles allows rapid gelation upon mixing, creating biomaterials with tailored properties. This reaction's speed and orthogonality position it as a valuable tool for in vivo applications and material science.
Sulfur(VI) Fluoride Exchange (SuFEx): An Emerging Click Reaction
SuFEx, a newer addition to the click chemistry arsenal, involves the exchange of sulfur(VI) fluorides with nucleophiles to form sulfonamide or sulfonate bonds. Driven by strong Si–F bond formation, it proceeds under mild, metal-free conditions with high stability and chemoselectivity. The review describes its application in synthesizing carbohydrate sulfonamides and multivalent architectures. Dondoni et al. prepared C-glucosylsulfonamides and calixarene-based clusters via SuFEx, demonstrating its potential for glycoconjugate functionalization. While still evolving, SuFEx complements other click reactions by offering unique bond-forming capabilities, particularly in protein modification and material science.
Connecting to GlycoCLICK™ Services at CD BioGlyco
The advancements detailed in this review align closely with the services offered by CD BioGlyco through our GlycoCLICK™ Platform. This suite leverages click chemistry principles—including metal-free variants—to support glycobiology research.
Our expertise in glycodendrimer synthesis and vaccine development resonates with the review's emphasis on multivalent systems and glycoconjugate vaccines. By offering tailored solutions, CD BioGlyco helps researchers overcome challenges in glycan-based therapeutic development, ensuring high-quality outcomes through advanced technological platforms.
Conclusion and Future Perspectives
The review by Ramírez-López et al. underscores the transformative impact of metal-free click reactions on glycochemistry and bioconjugation. TEC, SPAAC, IEDDA, and SuFEx provide robust, biocompatible tools for synthesizing complex glycoconjugates, enabling breakthroughs in biomedicine and materials science. As these technologies mature, integration with commercial services like CD BioGlyco's GlycoCLICK™ will accelerate innovation, offering researchers access to specialized expertise and resources. For those interested in exploring these applications further, visiting the GlycoCLICK™ services page can provide valuable insights and support for your projects. Embrace the future of glycobiology by leveraging these cutting-edge approaches to drive your research forward.
Reference
- Ramírez-López, P.; et al. Exploring metal-free click reactions: new frontiers in glycochemistry and bioconjugation. Bioconjugate Chemistry. 2025, 36(8): 1553-1581. (Open Access)

 Fig. 1. Comparative analysis of metal-free click reactions. (Ramírez-López, et al., 2025)
Fig. 1. Comparative analysis of metal-free click reactions. (Ramírez-López, et al., 2025)