Ultrafast Biomolecular Labeling via Photo-click Glycosylation
November 18, 2025
In a pioneering study titled "Photoactivatable electrophilic glycosylselenosulfonates for ultrafast modification of alkynes and thiols" published in ACS Central Science (2025), researchers from Fuzhou University, led by Yan et al. introduce a transformative class of bench-stable reagents that address critical limitations in glycosylseleno conjugate applications, such as slow reaction kinetics and poor functional group tolerance. By designing glycosylselenosulfonates with a unique dual-reactivity profile—combining electrophilic properties for rapid thiol modification and radical reactivity under mild photoirradiation for alkyne functionalization—the study achieves unprecedented reaction times of under one minute while maintaining high chemoselectivity and biocompatibility. This innovation enables efficient labeling of complex biomolecules, including insulin and cysteine-rich peptides, showcasing its potential to revolutionize real-time bioconjugation strategies for chemical biology and therapeutic development.
The Glycosylseleno Advantage and the Existing Challenge
Glycosylseleno conjugates, where a selenium atom replaces the oxygen at the sugar's anomeric center, are exceptionally valuable tools. Their unique properties make them ideal for multidisciplinary applications:
- Advanced structural analysis: The 77Se isotope serves as a powerful NMR-active nucleus for detailed structural studies, while selenium's heavy-atom effect enhances the resolution of X-ray crystallography for carbohydrate-protein complexes.
- Enhanced metabolic stability: These conjugates are resistant to glycosidase enzymes, making them promising candidates for stable therapeutic agents with high bioavailability.
- Unique spatial geometry: The distinct bond lengths and angles of selenium result in structures that can enable specific binding interactions not possible with their oxygen-based counterparts.
Despite these advantages, the synthesis and application of glycosylseleno scaffolds have been hampered by the limitations of existing reagents, which often suffer from slow reaction kinetics, poor efficiency, and limited functional group tolerance, rendering them unsuitable for real-time biological applications.
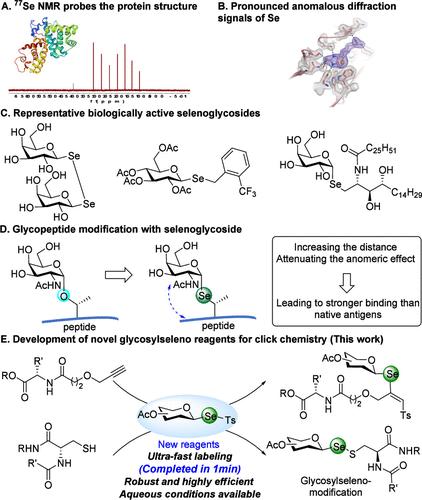 Fig.1 The landscape of glycosylseleno conjugate applications. (Yan, et al., 2025)
Fig.1 The landscape of glycosylseleno conjugate applications. (Yan, et al., 2025)
A Leap Forward: The Glycosylselenosulfonate Reagent
The research team, led by Professors Chunfa Xu and Ruo Wang, designed a novel solution to these limitations. They developed a bench-stable reagent class called glycosylselenosulfonates. The key innovation lies in replacing the carbon-centered leaving group found in traditional reagents with a sulfonyl group.
This strategic change offers a dual-reactivity profile:
- Electrophilic property: The polarized Se–SO2 bond facilitates rapid reactions with nucleophiles like thiols.
- Radical reactivity: The low bond dissociation energy (BDE) of the Se–S bond allows for clean homolytic cleavage under mild photoirradiation, generating reactive radicals.
This unique integration of radical and electrophilic properties in a single reagent is the cornerstone of its exceptional performance.
Demonstrated Capabilities: Ultrafast and Robust Reactions
The study showcases the remarkable capabilities of these reagents through two primary transformations:
- Photocatalytic Radical Addition to Alkynes
Upon irradiation with blue light in the presence of a ruthenium-based photocatalyst, glycosylselenosulfonates undergo a radical addition reaction with alkynes. This process is exceptionally fast, achieving completion in less than one minute. The reaction exhibits broad substrate scope, tolerating a wide range of functional groups (halogens, aldehydes, heteroaryl rings) and is compatible with various sugar donors (glucose, galactose, maltose). The method was successfully applied to label alkyne-functionalized amino acids and even the biomacromolecule insulin, highlighting its potential for complex biomolecule modification.
- Rapid Modification of Thiols (e.g., Cysteine Residues)
Demonstrating their pronounced electrophilicity, these reagents efficiently modify thiol-containing molecules, such as cysteine and cysteine-rich peptides like oxytocin. These reactions also proceed within approximately one minute under mild, dark conditions in aqueous-friendly solvent systems. Critically, the method shows excellent chemoselectivity for thiols over other amino acid side chains, a vital requirement for precise bioconjugation.
The robustness of both reactions was confirmed through additive experiments, where the presence of various amino acids and functional groups caused negligible interference, underscoring their potential for use in complex biological environments.
Bridging Cutting-Edge Research to Practical Application with CD BioGlyco
The development of glycosylselenosulfonates represents a significant academic achievement. However, leveraging such advanced chemical tools for practical drug discovery and biotechnology projects requires specialized expertise and infrastructure. This is where CD BioGlyco and its proprietary GlycoCLICK™ technology platform become an indispensable partner for researchers worldwide.
The ultrafast and selective nature of glycosylselenosulfonates aligns perfectly with the services offered by CD BioGlyco. Our platform is designed to help scientists exploit these very innovations for their specific goals:
We can utilize these novel reagents for the site-specific selenosylation of therapeutic proteins and antibodies, potentially enhancing their stability, solubility, or targeting capabilities.
Our expertise is ideal for applying this technology to label cell surfaces, specific biomolecules, or to develop novel fluorescent probes and biosensors based on the unique properties of the selenoglycoside linkage.
The rapid kinetics and biocompatibility of this chemistry make it suitable for constructing complex glycoconjugates for vaccine development or creating targeted drug delivery systems.
We can assist in functionalizing nanoparticles, liposomes, or hydrogels with glycosylseleno moieties to create advanced materials for diagnostic or therapeutic applications.
At CD BioGlyco, we combine deep expertise in glycochemistry with state-of-the-art technological platforms. Our team of specialists is ready to collaborate with you to design and execute a customized research plan, overcoming the technical challenges associated with implementing novel methodologies like glycosylselenosulfonate chemistry.
Reference
- Yan, W.; et al. Photoactivatable electrophilic glycosylselenosulfonates for ultrafast Modification of alkynes and thiols. ACS Central Science. 2025, 11(8): 1400-1407. (Open Access)

 Fig.1 The landscape of glycosylseleno conjugate applications. (Yan, et al., 2025)
Fig.1 The landscape of glycosylseleno conjugate applications. (Yan, et al., 2025)